Dr Healy and members of the MHRA met on 26 June 2009, the meetings purpose was to “discuss awareness and management of withdrawal reactions with SSRIs and related antidepressants”. The MHRA, Dr David Healy meetings minutes can be downloaded here.
Following the MHRAs receipt of and the documents eventual publication on the internet, it was found to include the statement that “Venlafaxine in doses up to 150 mg is an SSRI.”. Clarification of venlafxines’ SSRI v SNRI classification was sought by way of a Freedom of Information (FoI) question and request sent to Simon Gregor, the MHRAs Director of Communications.
The FoI request for clarification was made because of concerns that the Defining Issue of the Seroxat Group Litigation Order No: 68, lodged at the High Courts by the Litigations Lead Solicitors Hugh James -
“Does Seroxat have a capacity to cause asverse effects consequent upon or following discontinuance (withdrawl) such as prevent or make more difficult the ability of users to discontinue, withdraw from or remain free from taking Seroxat to a greater extent than all other Selective Serotonin Re-uptake Inhibitors (SSRIs)”is quite clear in it’s specification that it has to be proven that the discontinuation (withdrawal) and remaining free from Seroxat is worse and more difficult than any other “SSRI” in the drug class.
.…….. NB - spelling errors as per original document.
HM Courts Service: Seroxat Group Litigation Order no: 68.
With a certain amount of knowledge and the suspicion that the MHRA were not mandated to receive or facilitate a patient treatment protocol -- which would more likely fall under the sphere of influence of the National Institute for Clinical Excellence (NICE) -- an additional questions were asked to clarify if the MHRA were in a position, or intending, to authorise the Dr David Healy “Halting SSRIs” document as an official withdrawal protocol.
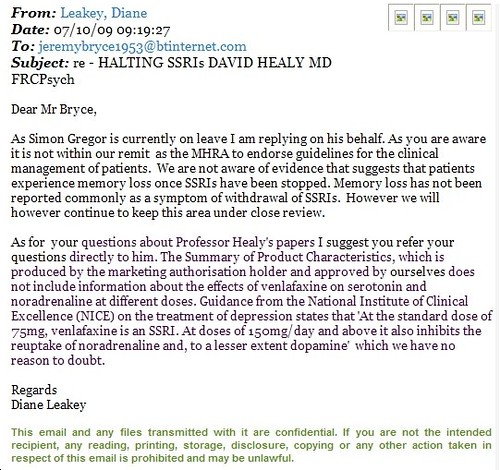
According the emailed reply (copy below) with regard the MHRA’s ability to facilitate utilising the Dr David Healy “Halting SSRIs” document the MHRA spokesperson states -
“As you are aware it is not within our remit to endorse guidelines for the management of patients.”With regard clarification of the venlafaxine SSRI v SNRI classification, the MHRA don’t appear to hold relatively basic information concerning the drugs dose related action on noradrenalin and serotonin –
although they had approved the “Summery of Product Characteristics, which is produced by the marketing authorisation holder” which presumably would have included the products name and category i.e. SSRI, SNRI, significance as such and dosage information.
The MHRA spokesperson did however quote from the official NICE “Guidance” on “treatment of depression” which states ‘At standard dose of 75 mg, venlafaxine is an SSRI. At doses of 150 mg/day and above it also inhibits the reuptake of noradrenaline and, to a lesser extent dopamine’ – adding the endorsement that they “have no reason to doubt” this.
Click here to enlarge
Published with permission of the recipient, original document here.
It would appear that the official view of the UK Governments Medicines and Healthcare products Regulatory Agency (MHRA), Department of Health (DH), National Health Service (NHS) and National Institute for Clinical Excellence (NICE) is that venlafaxine (Wyeth, Efexor UK, Effexor USA) , at standard doses up to 150 mg, is classified as an SSRI.

Personally speaking, Venlafaxine is an evil brain meddling, psychotic inducing, twisted thoughts inducing, family ruining and emotion deleting 'medicine'. What gives, those like me, (victims known as patients) NOTHING by way of, warning, professional assistance, ability to quit, nor a reasoning of the nonsensical 'marketing'. MHRA. What is it with the cluelessness in regards to your marketing approval, when the method of direction to affect the brain as with, clearly 'mentally damaging and lethal' Seroxat, is still there with all brain drugs? Venlafaxine, fluoxotine... Etc etc, are the same, they are nasty, they are not ever higher in benefit to the risks, the sinister and blatantly person changers, cause dysfunctions on the physical and mental once 'normalities' but how can this be happening?. (i learnt the hard way, Drs blindly worship Science, Medicinal Science knows sod all about the brain and cns, but wants to and theres only one way to learn). Worringly profit is so high and to sell, sell, sell along come the 'cheaper generics' that has to be said not all are so bioequivilent, nor use same inks on caps which renders any allergic reaction to, unidentifiable. The irony of it all??? Is very much 'we' as the public are educated from an early age MEDICINE IS GOOD FOR YOU. We accept to try the new cure (albeit no cures have been medication related despite it all) Why is a drug so damning on the market?
ReplyDeleteNote. No one replied to former post, i wonder why? Maybe, just maybe? It boils down to the ever simplistic 'in yer face' attitude that speaks truth is so hard to deny?, or that of arguement is futile when an expert really is, 'an expert'. due to reality of, 'being the drugged' and not, the actual quack phony, 'self acclaimed' expert via doing the drugging.
ReplyDeleteYou do realise people are seeing through the 'science of medicine' do you not?
Come on, face it, legal chemical drug pushing, killing and deforming can only be claimed as 'unknown underlying/unknown genetic/rare reaction' bull for a certified amount of time prior to the clicking and seeing exactly whats what.
Medication is very sinister, hasnt it always been?, 200yrs ago saw grave robbing, 100yrs ago saw new disease, disorder, dysfunction, whilst also new vaccine introductions, wonder drugs too, its now being said to drug babies at birth, and identifying certain genes that can be drugged to correct before take forms of mental disorder.
Brutality at its best but lets speak a truth here, whats the actual disease of depression, what causes it?, what cures it?, how does the cure work?, when is it noted asa cured?, when is it considered major? for what reason,?
Everyone suffers sadness, low periods, despair etc based on personal experience, answer this...... Two people exact same life style and circumstances, one is low as lack of finances the other is carefree and optimistic, how do the diferences pan out?, how can this be?, what if the optimistc one sees an ex with a new partner? What if that causes suicidal intent? The truth is nothing can determine a reaction in life, nothing can balance the diference. Depression is a fake diagnosis, so are all brain illnesses of the last decade or so, adhd, gad, sad etc, its pHARMing humans for profit and evil, drugs are bad unless science says so, science said of bad drugs today were scientifically good once.
Thalidamide was too obvious so its now best to go with brain mutation. Emotion is not disease, being low in this world is not abnormal, drugging people with chemical toxins claiming 'good therapy' is very abnormal, it makes you wonder how damaged a brain must be to believe in and want to do to others.? Emotions marketed as disease is due its karma, its a twisted and evil or brainwashed imbecile to not see the damage being done
Its the responsibility of the medicine maker that they should check its effects and then provide it in market.Celexa Birth Defects Lawsuit Attorney
ReplyDelete